A growing body of research suggests a link between chronic periodontal disease and Alzheimer's disease, centered on Porphyromonas gingivalis and its toxic enzymes called gingipains. While the findings are compelling, it is critical to understand that this research is still in its early stages. Correlation has been established, but causation has not been proven. What we can say with confidence is that maintaining good oral health may be one modifiable factor in a larger picture of brain health.
In 2019, a study published in Science Advances made headlines around the world: researchers had found Porphyromonas gingivalis — one of the primary bacteria responsible for chronic gum disease — inside the brains of Alzheimer's patients. The finding was startling, provocative, and immediately controversial.
Five years later, the science has evolved considerably. New studies have added nuance, some early therapeutic approaches have been tested (and some have failed), and the scientific community remains cautiously interested. Here is a measured look at what we actually know, what remains uncertain, and what — if anything — you should do about it.
The P. gingivalis Discovery
Porphyromonas gingivalis is an anaerobic bacterium that thrives in the deep pockets between teeth and gums in people with periodontitis. It is one of the so-called "red complex" pathogens and plays a central role in the destruction of gum tissue, bone loss, and tooth loss in advanced gum disease.
The landmark 2019 study by Dominy et al., published in Science Advances, reported three key findings:
- P. gingivalis DNA was identified in brain tissue from patients who had died with Alzheimer's disease, at significantly higher levels than in control brains.
- Gingipains — toxic protease enzymes produced by P. gingivalis — were detected in brain neurons and correlated with tau and ubiquitin pathology, two hallmarks of Alzheimer's.
- In a mouse model, oral infection with P. gingivalis led to brain colonization, increased amyloid-beta production, and neurodegeneration. A small-molecule gingipain inhibitor (COR388) reduced brain bacterial load and neuroinflammation.
The study proposed a provocative hypothesis: that P. gingivalis might not just be a bystander in Alzheimer's disease, but could play a causal role by migrating from the mouth to the brain and driving neurodegeneration through gingipain-mediated damage.
The Gingipain Hypothesis
Gingipains are cysteine proteases that P. gingivalis uses to break down host tissues in the gums. The Dominy study suggested these same enzymes could damage neurons once the bacteria reached the brain.
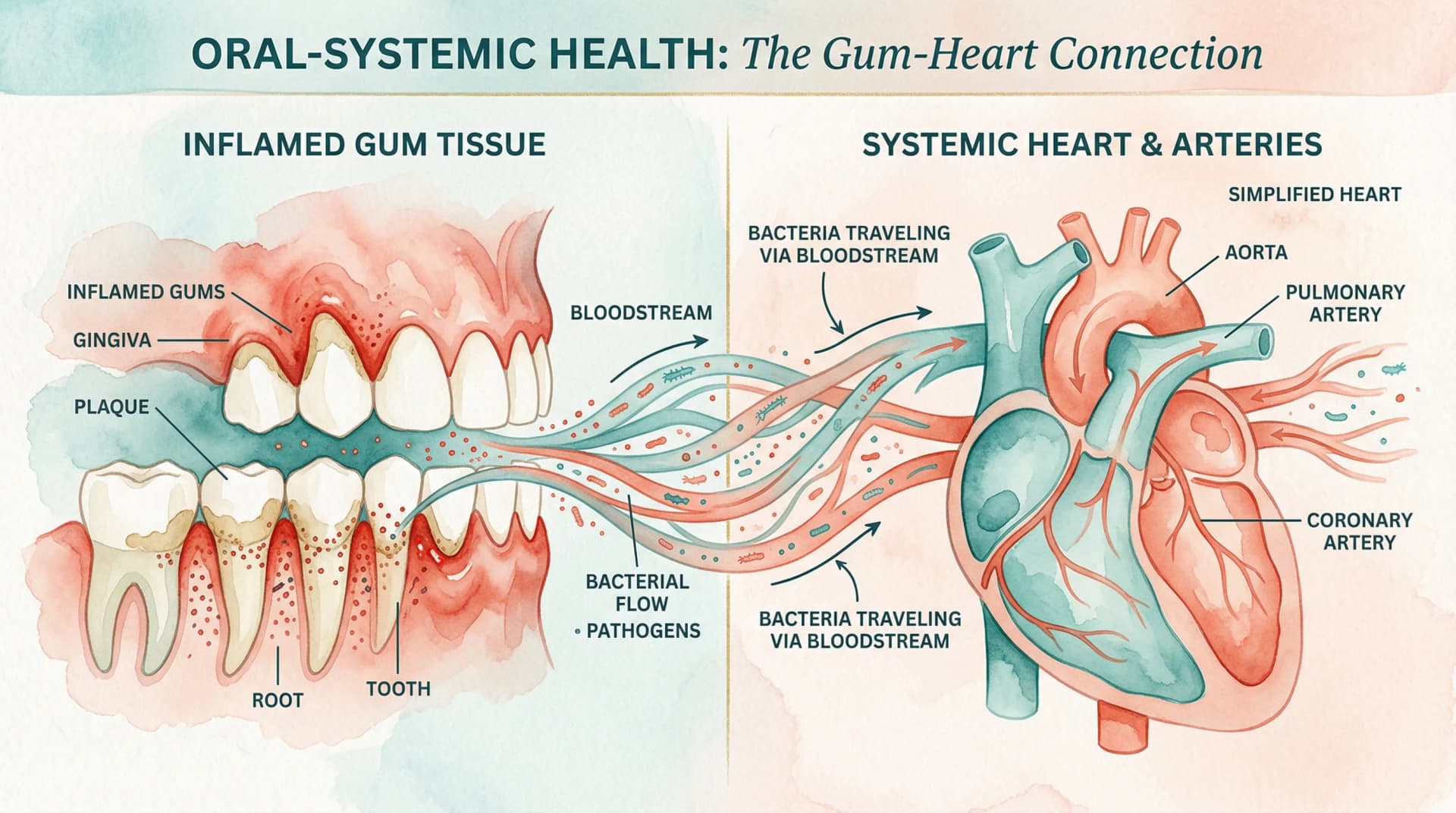
The proposed mechanism works roughly like this: chronic periodontal infection allows P. gingivalis to enter the bloodstream (bacteremia during chewing, brushing, or dental procedures is well documented). From the bloodstream, the bacteria may cross the blood-brain barrier — potentially facilitated by the chronic inflammation that both periodontitis and aging can cause — and establish colonies in brain tissue.
Once in the brain, gingipains could cleave tau proteins (contributing to neurofibrillary tangles), trigger amyloid-beta production as a defensive antimicrobial response, and activate neuroinflammatory cascades. If true, this would reframe Alzheimer's disease — at least in some patients — as partly driven by a chronic oral infection.
This hypothesis connects to the broader understanding of how gum disease affects the entire body. The mouth-body connection is well established for cardiovascular disease, and the potential brain connection follows a similar biological logic.
What Happened to COR388 (Atuzaginstat)?
One of the most watched developments was the clinical testing of COR388 (later named atuzaginstat), the gingipain inhibitor developed by Cortexyme, Inc. If the gingipain hypothesis was correct, blocking these enzymes should slow cognitive decline.
A Phase 2/3 clinical trial (the GAIN trial) enrolled over 600 patients with mild to moderate Alzheimer's. Results announced in late 2021 were disappointing: atuzaginstat did not meet its primary endpoints for slowing cognitive decline, as reported in JAMA Network Open.
However, the story was complicated by several factors. Post-hoc analyses suggested possible benefits in a subgroup of patients who had confirmed P. gingivalis infection (as measured by salivary gingipain levels), though subgroup analyses must be interpreted with great caution. Cortexyme also faced unrelated corporate governance issues that disrupted its research program.
The failure of atuzaginstat did not disprove the P. gingivalis hypothesis — it may simply mean that blocking gingipains alone is insufficient, that the drug was tested too late in the disease course, or that the hypothesis applies to only a subset of Alzheimer's patients. But it did temper some of the initial enthusiasm.
Correlation vs. Causation: The Critical Distinction
This is where intellectual honesty matters most. The epidemiological evidence linking periodontal disease and Alzheimer's is reasonably strong. A 2020 meta-analysis in the Journal of Alzheimer's Disease that pooled data from multiple cohort studies found that people with chronic periodontitis had a statistically significant increased risk of developing dementia.
But correlation is not causation, and there are several alternative explanations:
- Reverse causation — People in the early stages of cognitive decline may neglect oral hygiene, leading to periodontal disease as a consequence, not a cause, of neurodegeneration.
- Shared risk factors — Chronic inflammation, smoking, diabetes, socioeconomic status, and poor healthcare access are risk factors for both periodontitis and Alzheimer's. The association may reflect these shared drivers rather than a direct causal pathway.
- Bidirectional relationship — It is also possible that the relationship runs in both directions, with each condition exacerbating the other without either being the root cause.
Understanding the difference between gingivitis and periodontitis is relevant here. The research specifically implicates advanced chronic periodontitis — not mild gum inflammation — in the potential Alzheimer's connection. Gingivitis, which is reversible, has not been linked to the same systemic risks.
What the Latest Research Adds
Since the original 2019 paper, several lines of evidence have continued to develop:
-
A 2023 longitudinal study in Neurology followed over 8,000 adults for 18 years and found that tooth loss (a proxy for past periodontal disease) was associated with faster cognitive decline, even after adjusting for cardiovascular risk factors, education, and other confounders.
-
A 2024 study in Brain, Behavior, and Immunity provided new mechanistic evidence, showing that P. gingivalis outer membrane vesicles could cross blood-brain barrier models in vitro and trigger inflammatory responses in microglial cells.
-
Research into the gut-mouth connection has also highlighted that oral bacteria, including periodontal pathogens, may influence systemic inflammation through the gut-oral-brain axis, adding another potential pathway for the mouth-brain connection.
Taken together, the evidence is suggestive but not definitive. The scientific consensus, as expressed in a 2025 review in Periodontology 2000, is that chronic periodontitis is a "plausible contributing factor" to Alzheimer's risk but that proving direct causation will require much larger longitudinal studies and better tools for detecting early brain infection.
What This Means for You
Given the current state of evidence, here is a balanced perspective on practical implications:
What you should do:
- Maintain excellent oral hygiene — brush twice daily, floss, and attend regular dental checkups. This is sound advice regardless of the Alzheimer's connection.
- Treat periodontal disease promptly. If your dentist diagnoses gingivitis or periodontitis, follow their treatment recommendations. Letting chronic gum infection fester for years is a risk factor you can control.
- Manage systemic inflammation through diet, exercise, and chronic disease management (especially diabetes, which accelerates both periodontitis and cognitive decline).
What you should not do:
- Panic. Having gum disease does not mean you will develop Alzheimer's. The research shows a statistical association, not an inevitable outcome.
- Self-treat with antibiotics or antimicrobial mouthwashes in an attempt to kill P. gingivalis. Disrupting the oral microbiome indiscriminately can cause more harm than good.
- Ignore conventional Alzheimer's risk reduction strategies (physical activity, cognitive engagement, cardiovascular health, sleep quality) in favor of focusing solely on oral health.
The Bigger Picture
The gum disease-Alzheimer's connection is part of a broader paradigm shift in medicine: the recognition that chronic infections and the microbiome influence diseases we once thought were entirely genetic or age-related. The mouth, as the gateway to the body, is increasingly understood as a window into — and possibly a contributor to — systemic health.
Whether P. gingivalis turns out to be a direct driver of Alzheimer's or merely an amplifier of existing pathology, the practical takeaway is the same: your oral health matters far beyond your teeth and gums. Taking care of your mouth may be one of the most accessible things you can do for your long-term health.