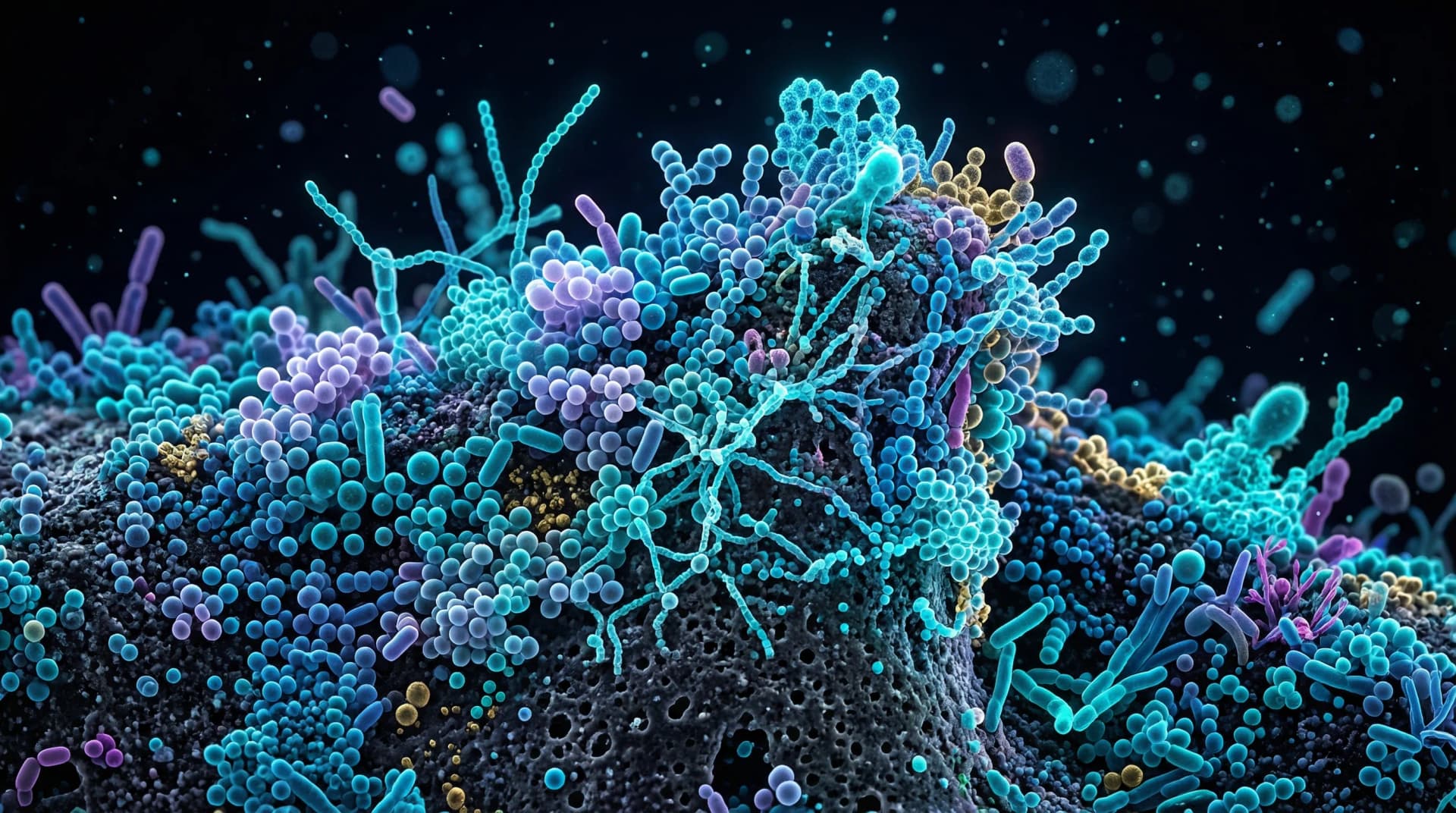
Antibiotics are essential medicines, but they don't discriminate between harmful and beneficial bacteria. A single course of broad-spectrum antibiotics can significantly disrupt the oral microbiome, reducing protective species and creating opportunities for opportunistic organisms like Candida to overgrow. Recovery may take weeks to months. Proactive steps — including oral probiotics, fermented foods, and careful oral hygiene — may help restore balance faster.
Antibiotics save lives. They've reduced mortality from bacterial infections to a fraction of what it was a century ago, and they remain one of modern medicine's most important tools.
But antibiotics have a well-known downside: they're indiscriminate. When you take amoxicillin for a sinus infection or azithromycin for bronchitis, the drug doesn't limit itself to the infection. It circulates throughout your body, reaching every microbial ecosystem — including the hundreds of species that make up your oral microbiome.
Understanding how antibiotics affect your mouth bacteria can help you take steps to protect your oral health during and after treatment.
How Antibiotics Disrupt the Oral Microbiome
Indiscriminate Killing
The oral microbiome exists in a state of dynamic balance. Beneficial species like Streptococcus sanguinis, Streptococcus gordonii, and various Veillonella species occupy ecological niches, compete with pathogens for resources, and help maintain a neutral or slightly alkaline oral pH.
Broad-spectrum antibiotics — the type most commonly prescribed — disrupt this balance by killing bacteria based on their structural characteristics (gram-positive vs. gram-negative, aerobic vs. anaerobic) rather than whether they're helpful or harmful to you.
A study published in mBio, 2019 used 16S rRNA sequencing to track oral microbiome changes in patients taking a one-week course of amoxicillin. The researchers found significant reductions in overall bacterial diversity, with some health-associated species dropping below detectable levels within just three days of antibiotic use.
Another study in the Journal of Oral Microbiology, 2018 found that a single course of broad-spectrum antibiotics reduced oral microbial diversity by an average of 25%, with certain protective species — notably Neisseria and Haemophilus — being disproportionately affected.
Loss of Colonization Resistance
One of the most important functions of a healthy oral microbiome is colonization resistance — the ability of established beneficial bacteria to prevent harmful organisms from gaining a foothold. When antibiotics deplete these protective species, they create open ecological niches that opportunistic organisms can exploit.
Research in Trends in Microbiology, 2016 described how antibiotics dismantle colonization resistance through three mechanisms:
- Direct elimination of protective species that compete with pathogens
- Reduced production of antimicrobial compounds (bacteriocins) that beneficial bacteria normally produce to suppress pathogens
- Altered nutrient availability — dead bacteria release nutrients that can fuel pathogen growth
The practical result is that your mouth becomes temporarily more vulnerable to infection and imbalance after antibiotic treatment.
Antibiotic-Resistant Survivors
Not all bacteria are equally susceptible to a given antibiotic. The species that survive a course of antibiotics — either through innate resistance or acquired resistance genes — can rapidly expand into the ecological space left by killed competitors.
A longitudinal study in Microbiome, 2019 found that oral microbial communities after antibiotic treatment were enriched in antibiotic-resistance genes, and these resistant populations persisted for months after treatment ended. This has implications not just for individual oral health but for the broader public health challenge of antibiotic resistance.
Increased Candida Risk: Oral Thrush
Perhaps the most visible oral side effect of antibiotic use is oral thrush — an overgrowth of the fungus Candida albicans that causes white, cottage cheese-like patches on the tongue, inner cheeks, and roof of the mouth.

Candida is a normal resident of the mouth in small numbers. Under healthy conditions, bacterial competitors keep it in check. When antibiotics eliminate those bacterial competitors, Candida can proliferate unchecked.
A study in the Journal of Antimicrobial Chemotherapy, 2003 found that oral Candida colonization increased significantly during antibiotic treatment, with rates jumping from approximately 30% to over 60% in hospitalized patients receiving broad-spectrum antibiotics.
Symptoms of Oral Thrush
- White or cream-colored patches on the tongue, cheeks, gums, or throat
- Redness or soreness underneath the patches
- Cotton-like sensation in the mouth
- Loss of taste or an unpleasant taste
- Pain when eating or swallowing
- Cracking at the corners of the mouth (angular cheilitis)
If you develop oral thrush during or after antibiotic treatment, see your dentist or doctor. Treatment typically involves antifungal medications like nystatin or fluconazole.
How Long Does Recovery Take?
The timeline for oral microbiome recovery after antibiotics varies depending on the type of antibiotic, duration of treatment, and individual factors. But research consistently shows that recovery takes longer than most people assume.
The Research on Recovery Timelines
A comprehensive study published in Nature Microbiology, 2018 tracked microbiome recovery in healthy volunteers after antibiotic exposure. While some bacterial populations began recovering within days of stopping antibiotics, the researchers found that:
- Partial recovery (return of most major species): 1-4 weeks
- Substantial recovery (community composition resembling pre-antibiotic state): 1-3 months
- Some species had not recovered to baseline levels even at 6 months post-treatment
The oral microbiome generally recovers faster than the gut microbiome, likely because the mouth is an open ecosystem constantly exposed to new bacteria from food, water, and the environment. However, full restoration of the pre-antibiotic community structure is not guaranteed — the recovered microbiome may be a "new normal" that differs from the original composition.
Factors That Influence Recovery Speed
- Antibiotic type: Narrow-spectrum antibiotics (targeting specific bacterial groups) cause less disruption than broad-spectrum ones
- Duration of treatment: Longer courses cause more damage and slower recovery
- Repeated courses: Multiple antibiotic courses within a short period compound the damage
- Diet during and after treatment: Nutrient-rich, fiber-heavy diets may support faster recovery
- Oral hygiene practices: Excessive use of antiseptic mouthwash during recovery can further delay restoration
- Age and overall health: Older adults and immunocompromised individuals may recover more slowly
How to Protect Your Oral Health During Antibiotic Treatment
If you need antibiotics — and sometimes you genuinely do — here are evidence-informed strategies to minimize oral microbiome disruption and support faster recovery.
1. Don't Skip Antibiotics You Truly Need
This is important: the goal is not to avoid antibiotics at all costs. Untreated bacterial infections can be dangerous. The goal is to protect your oral microbiome while taking necessary antibiotics, and to avoid unnecessary antibiotic prescriptions.
If your doctor prescribes antibiotics, take the full course as directed. Stopping early doesn't "save" your microbiome — it increases the risk of antibiotic resistance and treatment failure.
2. Ask About Narrow-Spectrum Options
When appropriate, ask your prescriber whether a narrow-spectrum antibiotic could be effective for your condition. Narrow-spectrum antibiotics target a more limited range of bacteria, potentially causing less collateral damage to your microbiome.
This isn't always possible — some infections require broad-spectrum coverage — but it's a reasonable question to raise.
3. Maintain Excellent Oral Hygiene
During antibiotic treatment, consistent brushing and flossing become even more important. With your microbiome disrupted, you want to physically remove plaque and food debris that could feed opportunistic organisms.
However, avoid antiseptic mouthwashes (like those containing chlorhexidine or cetylpyridinium chloride) during the recovery period unless specifically prescribed by your dentist. These products further reduce bacterial populations at a time when you're trying to rebuild them.
4. Eat to Support Microbiome Recovery
Diet plays a meaningful role in microbiome recovery. During and after antibiotic treatment:
- Increase fermented food intake: Yogurt with live cultures, kefir, sauerkraut, and kimchi introduce beneficial bacteria. A study in Cell, 2021 found that a fermented-food-rich diet increased microbial diversity more effectively than a high-fiber diet alone.
- Eat prebiotic-rich foods: Garlic, onions, bananas, asparagus, and oats contain fibers that nourish beneficial bacteria.
- Reduce sugar: With colonization resistance compromised, sugar has an even greater ability to fuel pathogenic overgrowth during antibiotic treatment.
- Stay hydrated: Water supports saliva production, which is critical for maintaining oral homeostasis.
5. Consider Oral Probiotics
Probiotic supplementation during and after antibiotic use is one of the most actively researched recovery strategies. The rationale is straightforward: if antibiotics deplete beneficial bacteria, reintroducing specific beneficial strains may help reestablish a healthier microbial balance.
A meta-analysis published in the Journal of the American Medical Association, 2012 found that probiotic use during antibiotic treatment reduced the risk of antibiotic-associated complications, particularly Clostridium difficile infection and diarrhea.
For oral-specific benefits, the strain Lactobacillus reuteri has the strongest evidence base. A study in the European Journal of Clinical Microbiology & Infectious Diseases, 2015 found that L. reuteri supplementation during antibiotic treatment helped preserve beneficial oral bacteria and reduced opportunistic pathogen colonization.

ProDentim
Oral Probiotic for Gum & Teeth Health
ProDentim contains Lactobacillus reuteri and other beneficial strains delivered directly to the oral cavity via a dissolving tablet — a targeted approach to supporting oral microbiome recovery during and after antibiotic courses.
We may earn a commission if you make a purchase through our links, at no extra cost to you.
6. Time Your Probiotics Correctly
If you're taking probiotics alongside antibiotics, timing matters. Take your probiotic dose at least 2-3 hours before or after your antibiotic dose. This gives the probiotic bacteria a window to establish themselves before the next antibiotic dose arrives.
Continue probiotic supplementation for at least 2-4 weeks after completing your antibiotic course to support ongoing recovery.
When to Consult Your Dentist
If you experience any of the following during or after antibiotic treatment, consult your dentist:
- White patches or persistent soreness in the mouth (possible thrush)
- Increased gum bleeding or swelling
- New or worsening bad breath that persists after antibiotics are finished
- Tooth sensitivity that wasn't present before treatment
- Any oral symptoms that don't resolve within a month of completing antibiotics
Your dentist can assess whether antibiotic-related microbiome disruption has led to conditions requiring treatment and recommend appropriate interventions.
Antibiotics in Dentistry: A Special Consideration
Dentists themselves prescribe antibiotics — for dental abscesses, pre-surgical prophylaxis, and aggressive periodontal infections. If your dentist prescribes antibiotics, the same microbiome concerns apply.
Research published in the Journal of the American Dental Association, 2019 noted that dental professionals account for approximately 10% of all outpatient antibiotic prescriptions in the United States. The profession has been actively working to reduce unnecessary prescriptions through antibiotic stewardship programs.
If your dentist recommends antibiotics, it's appropriate to ask:
- Is this antibiotic necessary, or are there alternative treatment approaches?
- Can a narrow-spectrum antibiotic be used?
- What's the minimum effective duration?
These are not confrontational questions — they align with current best practice in antibiotic stewardship.
The Long View: Building Resilience
The best protection against antibiotic-related microbiome disruption is a robust, diverse microbiome going in. Like a diverse ecosystem in nature, a diverse oral microbiome is more resilient to disturbance and recovers faster.
You can build oral microbiome resilience through:
- A varied, whole-foods diet rich in fiber and polyphenols — see our guide to foods that support gum health
- Regular consumption of fermented foods
- Avoiding unnecessary antimicrobial products (antiseptic mouthwash for daily use)
- Good oral hygiene that controls plaque without sterilizing the mouth
- Oral probiotic supplementation as part of your routine

ProDentim
Oral Probiotic for Gum & Teeth Health
Regular oral probiotic use may help maintain a diverse oral microbiome that's better equipped to withstand disruptions from antibiotic treatment. ProDentim's multi-strain formula is designed specifically for oral colonization.
We may earn a commission if you make a purchase through our links, at no extra cost to you.
Not every antibiotic course will cause noticeable oral problems. Many people take antibiotics and experience no obvious changes in their mouth. But the microbiome disruption is happening beneath the surface regardless, and taking proactive steps to support recovery is a worthwhile investment in your long-term oral health.
Related reading:
- mouthwash kills good bacteria too
- oral probiotics for recovery
- Lactobacillus reuteri for oral health